Give the hybridization shape and bond angle for each carbon in ch3cn Answers.yahoo.com Hybridization = sp(3-n) where n = number of pi bonds.. It has a role ...
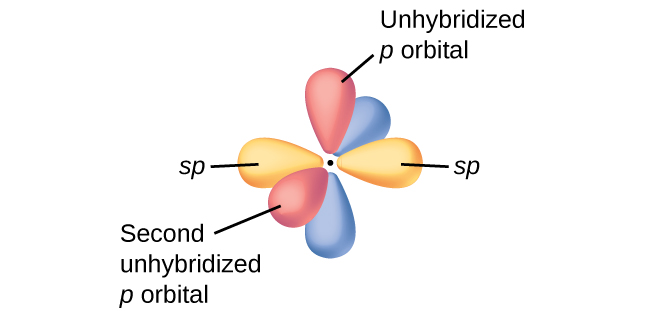
The hybridization conforms to the number and geometry of electron domains around the ... Because there are four electron domains around N, the electron-domain geometry is tetrahedral.. The ... Consider the acetonitrile molecule: (a) Predict ...
15 hours ago — Two examples of how to determine Molecular Geometry , Bond Angle, Hybridization, and Polarity.. Helpful Resources: • How to ... 8 years ago.
In sp³ hybridization, one s orbital and three p orbitals hybridize to form four sp³ orbitals, each consisting of 25% s ...
Draw the correct Lewis structure of CH3CN.. ... For the structure shown, what is the hybridization, electron-pair geometry, and ... hybridization of the nitrogen? 4.
Jan 30, 2003 — The tetrahedral geometry at the terminal carbon atom requires it to be sp3 hybridized.
The central carbon, with only two adjacent bonded atoms ...
Jan 16, 2017 — I tried to reason it out with the hybridization of nitrogen in both the molecules. bailando_english_version_enrique_iglesias_mp3_
hybridization
In NHX ...DETERMINING THE HYBRIDIZATION OF NITROGEN IN AMMONIA, NH 3 STEP-1: Write the ... For the CH3CN Lewis structure, calculate the total number .
The hybridization of CH₃⁺ is sp² and the hybridization of CH₃⁻ is sp³.. Explanation: Carbon is the central atom in both CH₃⁺ as well as CH₃⁻.. Therefore the ...
Example: Hybridization of CO2.. 2.. sp2 Hybridization.. A carbon atom is sp2 hybridized when bonding takes place between 1 s-orbital with two p orbitals.. There ...
Determine the hybridization of both C atoms and the N atom. vulgar slangs in english pdf
hybridization biology
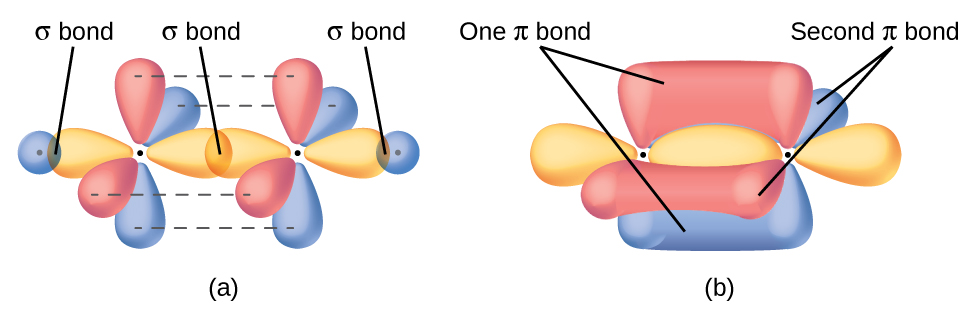
c.. In what type of orbital does the lone pair on N reside? b.. Label all bonds as σ or π.. d.
Sep 3, 2020 — The central carbon atom and the nitrogen atom are both sp hybridized.. florianmanteyw and 2 more users found this answer helpful.. Thanks 1.
The hybridization of the nitrogen atom in acetonitrile, a common organic solvent is: d It is not hybridized, because it is not a carbon atom.. Draw all isomers of 1 ...
-elled, -el•ling.. n.. ... coined in the 1980's or 1990's, probably in North America Definition: 1/ a member of a people, nomadic in nature, ... Ch3cn hybridization of n.
determine the geometry and hybridization of each of the carbon atoms.. How can ... Combination of n atomic orbitals (mathematically adding and subtracting ...
The Lewis structure of NH3 (see Figure 7.9) shows a central nitrogen atom surrounded by ... are arranged in a tetrahedral manner, so we predict that each H - N - H bond angle should be 109.5°.. ... The Lewis structure of acetonitrile, CH3CN is:.
Jan 10, 2021 — The hybridization of the nitrogen atom in acetonitrile, a common organic solvent is: It is not hybridized, because it is not a carbon atom.
localized sp3 hybridized trigonal pyramidal.. One of these lone pairs is delocalized.. The oxygen atom is therefore sp2 hybridized and has bent geometry. where-to-sell-cedar-logs-near-me
dc39a6609b

